I Conducted a Search for Meta-analyses and Systematic Reviews
- Review
- Open Admission
- Published:
A step by footstep guide for conducting a systematic review and meta-analysis with simulation information
Tropical Medicine and Health volume 47, Article number:46 (2019) Cite this article
Abstract
Groundwork
The massive affluence of studies relating to tropical medicine and health has increased strikingly over the concluding few decades. In the field of tropical medicine and health, a well-conducted systematic review and meta-analysis (SR/MA) is considered a feasible solution for keeping clinicians abreast of current evidence-based medicine. Agreement of SR/MA steps is of paramount importance for its conduction. It is non like shooting fish in a barrel to be done every bit in that location are obstacles that could face the researcher. To solve those hindrances, this methodology study aimed to provide a step-by-footstep approach mainly for beginners and inferior researchers, in the field of tropical medicine and other health care fields, on how to properly conduct a SR/MA, in which all the steps hither depicts our experience and expertise combined with the already well-known and accepted international guidance.
We propose that all steps of SR/MA should exist done independently past 2–3 reviewers' discussion, to ensure data quality and accuracy.
Decision
SR/MA steps include the evolution of research question, forming criteria, search strategy, searching databases, protocol registration, title, abstract, full-text screening, transmission searching, extracting data, quality assessment, information checking, statistical analysis, double information checking, and manuscript writing.
Introduction
The amount of studies published in the biomedical literature, especially tropical medicine and health, has increased strikingly over the last few decades. This massive abundance of literature makes clinical medicine increasingly complex, and noesis from various researches is oft needed to inform a particular clinical decision. However, available studies are often heterogeneous with regard to their blueprint, operational quality, and subjects under study and may handle the research question in a different way, which adds to the complexity of evidence and determination synthesis [i].
Systematic review and meta-analyses (SR/MAs) have a high level of evidence as represented past the prove-based pyramid. Therefore, a well-conducted SR/MA is considered a feasible solution in keeping health clinicians ahead regarding contemporary testify-based medicine.
Differing from a systematic review, unsystematic narrative review tends to be descriptive, in which the authors select often articles based on their point of view which leads to its poor quality. A systematic review, on the other hand, is defined every bit a review using a systematic method to summarize evidence on questions with a detailed and comprehensive programme of study. Furthermore, despite the increasing guidelines for effectively conducting a systematic review, we found that basic steps oft start from framing question, then identifying relevant work which consists of criteria development and search for articles, assess the quality of included studies, summarize the evidence, and interpret the results [2, 3]. All the same, those simple steps are not easy to be reached in reality. There are many troubles that a researcher could be struggled with which has no detailed indication.
Conducting a SR/MA in tropical medicine and health may be difficult especially for young researchers; therefore, understanding of its essential steps is crucial. It is not easy to exist done as there are obstacles that could confront the researcher. To solve those hindrances, we recommend a flow diagram (Fig. 1) which illustrates a detailed and step-by-pace the stages for SR/MA studies. This methodology report aimed to provide a step-by-step arroyo mainly for beginners and inferior researchers, in the field of tropical medicine and other health care fields, on how to properly and succinctly carry a SR/MA; all the steps here depicts our experience and expertise combined with the already well known and accepted international guidance.
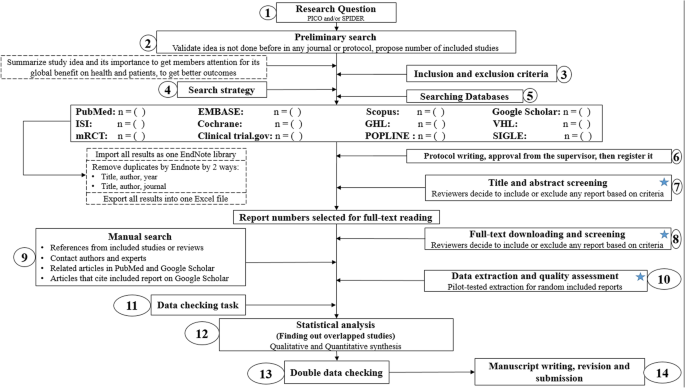
Detailed flow diagram guideline for systematic review and meta-assay steps. Annotation: Star icon refers to "2–3 reviewers screen independently"
Methods and results
Detailed steps for conducting whatever systematic review and meta-analysis
We searched the methods reported in published SR/MA in tropical medicine and other healthcare fields too the published guidelines like Cochrane guidelines {Higgins, 2011 #7} [four] to collect the best low-bias method for each footstep of SR/MA conduction steps. Furthermore, we used guidelines that we apply in studies for all SR/MA steps. Nosotros combined these methods in order to conclude and conduct a detailed flow diagram that shows the SR/MA steps how existence conducted.
Whatever SR/MA must follow the widely accepted Preferred Reporting Items for Systematic Review and Meta-analysis statement (PRISMA checklist 2009) (Additional file v: Table S1) [five].
We proposed our methods co-ordinate to a valid explanatory simulation example choosing the topic of "evaluating safety of Ebola vaccine," as it is known that Ebola is a very rare tropical illness just fatal. All the explained methods feature the standards followed internationally, with our compiled experience in the conduct of SR beside information technology, which we call up proved some validity. This is a SR under conduct by a couple of researchers teaming in a inquiry group, moreover, as the outbreak of Ebola which took place (2013–2016) in Africa resulted in a significant mortality and morbidity. Furthermore, since at that place are many published and ongoing trials assessing the condom of Ebola vaccines, we idea this would provide a great opportunity to tackle this hotly debated effect. Moreover, Ebola started to burn down again and new fatal outbreak appeared in the Democratic Democracy of Congo since August 2018, which caused infection to more than than g people according to the Earth Health System, and 629 people accept been killed till now. Hence, it is considered the second worst Ebola outbreak, after the first ane in West Africa in 2014, which infected more than than 26,000 and killed most 11,300 people along outbreak grade.
Enquiry question and objectives
Like other written report designs, the research question of SR/MA should be feasible, interesting, novel, ethical, and relevant. Therefore, a clear, logical, and well-defined inquiry question should exist formulated. Usually, 2 common tools are used: PICO or SPIDER. PICO (Population, Intervention, Comparison, Result) is used more often than not in quantitative evidence synthesis. Authors demonstrated that PICO holds more sensitivity than the more specific SPIDER approach [6]. SPIDER (Sample, Phenomenon of Interest, Blueprint, Evaluation, Research blazon) was proposed as a method for qualitative and mixed methods search.
We here recommend a combined approach of using either one or both the SPIDER and PICO tools to call up a comprehensive search depending on time and resources limitations. When nosotros apply this to our assumed enquiry topic, existence of qualitative nature, the use of SPIDER approach is more than valid.
PICO is usually used for systematic review and meta-analysis of clinical trial written report. For the observational written report (without intervention or comparator), in many tropical and epidemiological questions, it is unremarkably plenty to use P (Patient) and O (event) only to formulate a research question. We must indicate clearly the population (P), so intervention (I) or exposure. Adjacent, it is necessary to compare (C) the indicated intervention with other interventions, i.e., placebo. Finally, we demand to clarify which are our relevant outcomes.
To facilitate comprehension, we choose the Ebola virus disease (EVD) as an example. Currently, the vaccine for EVD is existence developed and under phase I, Two, and III clinical trials; we desire to know whether this vaccine is safe and can induce sufficient immunogenicity to the subjects.
An example of a research question for SR/MA based on PICO for this issue is every bit follows: How is the prophylactic and immunogenicity of Ebola vaccine in human? (P: healthy subjects (human), I: vaccination, C: placebo, O: safe or adverse effects)
Preliminary research and idea validation
Nosotros recommend a preliminary search to identify relevant articles, ensure the validity of the proposed idea, avoid duplication of previously addressed questions, and clinch that we have enough articles for conducting its assay. Moreover, themes should focus on relevant and important health-care issues, consider global needs and values, reflect the current science, and exist consistent with the adopted review methods. Gaining familiarity with a deep understanding of the study field through relevant videos and discussions is of paramount importance for ameliorate retrieval of results. If we ignore this step, our study could be canceled whenever we find out a similar study published before. This means nosotros are wasting our fourth dimension to deal with a problem that has been tackled for a long time.
To exercise this, we can start past doing a unproblematic search in PubMed or Google Scholar with search terms Ebola AND vaccine. While doing this stride, nosotros identify a systematic review and meta-analysis of determinant factors influencing antibody response from vaccination of Ebola vaccine in not-human primate and human [seven], which is a relevant newspaper to read to go a deeper insight and place gaps for better formulation of our enquiry question or purpose. We tin can notwithstanding conduct systematic review and meta-analysis of Ebola vaccine because nosotros evaluate condom as a different outcome and different population (merely human being).
Inclusion and exclusion criteria
Eligibility criteria are based on the PICO approach, study design, and date. Exclusion criteria mostly are unrelated, duplicated, unavailable total texts, or abstruse-only papers. These exclusions should be stated in advance to refrain the researcher from bias. The inclusion criteria would exist articles with the target patients, investigated interventions, or the comparison between 2 studied interventions. Briefly, it would be manufactures which contain information answering our research question. Only the well-nigh important is that information technology should be articulate and sufficient data, including positive or negative, to answer the question.
For the topic we have chosen, we tin make inclusion criteria: (one) whatsoever clinical trial evaluating the safety of Ebola vaccine and (2) no restriction regarding state, patient age, race, gender, publication language, and date. Exclusion criteria are equally follows: (1) report of Ebola vaccine in non-human subjects or in vitro studies; (2) study with information non reliably extracted, indistinguishable, or overlapping data; (3) abstract-only papers as preceding papers, conference, editorial, and author response theses and books; (four) articles without available full text available; and (v) case reports, case series, and systematic review studies. The PRISMA flow diagram template that is used in SR/MA studies tin be found in Fig. 2.
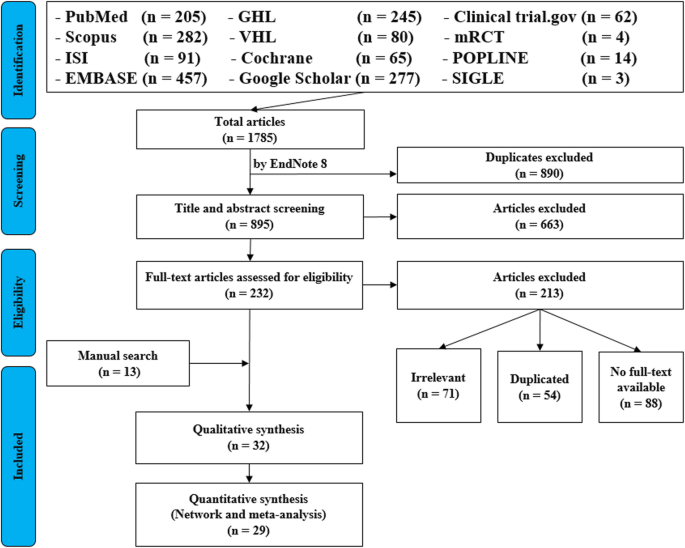
PRISMA menstruation diagram of studies' screening and selection
Search strategy
A standard search strategy is used in PubMed, and then afterwards information technology is modified co-ordinate to each specific database to get the best relevant results. The basic search strategy is congenital based on the research question conception (i.e., PICO or PICOS). Search strategies are synthetic to include costless-text terms (e.g., in the title and abstract) and any appropriate field of study indexing (e.g., MeSH) expected to recollect eligible studies, with the help of an expert in the review topic field or an information specialist. Additionally, we propose not to use terms for the Outcomes every bit their inclusion might hinder the database existence searched to recall eligible studies considering the used outcome is not mentioned evidently in the articles.
The improvement of the search term is made while doing a trial search and looking for some other relevant term within each concept from retrieved papers. To search for a clinical trial, we can apply these descriptors in PubMed: "clinical trial"[Publication Type] OR "clinical trials as topic"[MeSH terms] OR "clinical trial"[All Fields]. After some rounds of trial and refinement of search term, we formulate the final search term for PubMed as follows: (ebola OR ebola virus OR ebola virus disease OR EVD) AND (vaccine OR vaccination OR vaccinated OR immunization) AND ("clinical trial"[Publication Blazon] OR "clinical trials as topic"[MeSH Terms] OR "clinical trial"[All Fields]). Because the written report for this topic is limited, we practice not include outcome term (safety and immunogenicity) in the search term to capture more studies.
Search databases, import all results to a library, and exporting to an excel sheet
According to the AMSTAR guidelines, at least 2 databases have to be searched in the SR/MA [viii], but as you increment the number of searched databases, you get much yield and more authentic and comprehensive results. The ordering of the databases depends generally on the review questions; being in a study of clinical trials, you lot volition rely mostly on Cochrane, mRCTs, or International Clinical Trials Registry Platform (ICTRP). Here, we propose 12 databases (PubMed, Scopus, Web of Science, EMBASE, GHL, VHL, Cochrane, Google Scholar, Clinical trials.gov, mRCTs, POPLINE, and SIGLE), which assist to cover nigh all published manufactures in tropical medicine and other health-related fields. Amid those databases, POPLINE focuses on reproductive wellness. Researchers should consider to choose relevant database according to the inquiry topic. Some databases do not support the use of Boolean or quotation; otherwise, there are some databases that have special searching mode. Therefore, we need to modify the initial search terms for each database to become appreciated results; therefore, manipulation guides for each online database searches are presented in Additional file 5: Table S2. The detailed search strategy for each database is found in Additional file five: Table S3. The search term that we created in PubMed needs customization based on a specific characteristic of the database. An case for Google Scholar avant-garde search for our topic is as follows:
- 1.
With all of the words: ebola virus
With at least one of the words: vaccine vaccination vaccinated immunization
Where my words occur: in the title of the commodity
- 2.
With all of the words: EVD
With at to the lowest degree one of the words: vaccine vaccination vaccinated immunization
Where my words occur: in the title of the article
Finally, all records are collected into one Endnote library in guild to delete duplicates then to it consign into an excel canvass. Using remove duplicating function with two options is mandatory. All references which have (1) the aforementioned championship and writer, and published in the same year, and (2) the same title and author, and published in the same journal, would be deleted. References remaining afterward this step should be exported to an excel file with essential data for screening. These could be the authors' names, publication year, journal, DOI, URL link, and abstract.
Protocol writing and registration
Protocol registration at an early stage guarantees transparency in the research process and protects from duplication problems. Besides, information technology is considered a documented proof of squad programme of action, research question, eligibility criteria, intervention/exposure, quality assessment, and pre-assay program. It is recommended that researchers send information technology to the main investigator (PI) to revise it, then upload it to registry sites. There are many registry sites available for SR/MA like those proposed by Cochrane and Campbell collaborations; nevertheless, we recommend registering the protocol into PROSPERO as it is easier. The layout of a protocol template, co-ordinate to PROSPERO, can be plant in Boosted file five: File S1.
Championship and abstract screening
Decisions to select retrieved manufactures for farther cess are based on eligibility criteria, to minimize the adventure of including non-relevant articles. According to the Cochrane guidance, two reviewers are a must to do this pace, but equally for beginners and junior researchers, this might be tiresome; thus, nosotros propose based on our experience that at least three reviewers should work independently to reduce the chance of mistake, specially in teams with a large number of authors to add more than scrutiny and ensure proper conduct. Mostly, the quality with three reviewers would be better than two, as ii only would have different opinions from each other, so they cannot make up one's mind, while the third opinion is crucial. And here are some examples of systematic reviews which we conducted following the aforementioned strategy (past a different grouping of researchers in our research grouping) and published successfully, and they feature relevant ideas to tropical medicine and illness [nine,10,xi].
In this step, duplications will exist removed manually whenever the reviewers detect them out. When there is a doubt nigh an article decision, the team should be inclusive rather than exclusive, until the main leader or PI makes a determination after discussion and consensus. All excluded records should be given exclusion reasons.
Total text downloading and screening
Many search engines provide links for gratis to admission full-text manufactures. In example non found, we can search in some research websites as ResearchGate, which offer an option of direct full-text request from authors. Additionally, exploring archives of wanted journals, or contacting PI to purchase it if available. Similarly, ii–3 reviewers work independently to decide about included full texts according to eligibility criteria, with reporting exclusion reasons of articles. In case any disagreement has occurred, the final decision has to exist fabricated by discussion.
Manual search
I has to exhaust all possibilities to reduce bias by performing an explicit hand-searching for retrieval of reports that may accept been dropped from first search [12]. Nosotros apply five methods to make manual searching: searching references from included studies/reviews, contacting authors and experts, and looking at related articles/cited articles in PubMed and Google Scholar.
We describe here iii consecutive methods to increase and refine the yield of manual searching: firstly, searching reference lists of included articles; secondly, performing what is known equally citation tracking in which the reviewers rail all the articles that cite each one of the included manufactures, and this might involve electronic searching of databases; and thirdly, similar to the citation tracking, we follow all "related to" or "similar" articles. Each of the abovementioned methods can be performed by 2–iii contained reviewers, and all the possible relevant article must undergo further scrutiny against the inclusion criteria, after post-obit the aforementioned records yielded from electronic databases, i.eastward., title/abstract and full-text screening.
We propose an contained reviewing past assigning each member of the teams a "tag" and a distinct method, to compile all the results at the end for comparison of differences and give-and-take and to maximize the retrieval and minimize the bias. Similarly, the number of included articles has to exist stated before improver to the overall included records.
Data extraction and quality assessment
This step entitles data drove from included total-texts in a structured extraction excel sheet, which is previously pilot-tested for extraction using some random studies. We recommend extracting both adjusted and non-adjusted information because it gives the nearly immune confounding factor to exist used in the analysis by pooling them later [13]. The procedure of extraction should be executed by 2–3 independent reviewers. Mostly, the sheet is classified into the study and patient characteristics, outcomes, and quality assessment (QA) tool.
Data presented in graphs should be extracted past software tools such as Web plot digitizer [14]. Most of the equations that tin be used in extraction prior to analysis and interpretation of standard deviation (SD) from other variables is found inside Boosted file five: File S2 with their references as Hozo et al. [15], Xiang et al. [16], and Rijkom et al. [17]. A variety of tools are available for the QA, depending on the pattern: ROB-2 Cochrane tool for randomized controlled trials [xviii] which is presented as Additional file 1: Figure S1 and Additional file 2: Figure S2—from a previous published article data—[19], NIH tool for observational and cross-sectional studies [20], ROBINS-I tool for not-randomize trials [21], QUADAS-two tool for diagnostic studies, QUIPS tool for prognostic studies, CARE tool for case reports, and ToxRtool for in vivo and in vitro studies. Nosotros recommend that ii–3 reviewers independently appraise the quality of the studies and add to the data extraction form earlier the inclusion into the analysis to reduce the risk of bias. In the NIH tool for observational studies—cohort and cantankerous-exclusive—as in this EBOLA case, to evaluate the risk of bias, reviewers should rate each of the 14 items into dichotomous variables: yes, no, or not applicable. An overall score is calculated by adding all the items scores as yeah equals ane, while no and NA equals nada. A score will exist given for every paper to classify them as poor, off-white, or good conducted studies, where a score from 0–5 was considered poor, 6–9 every bit fair, and 10–fourteen as good.
In the EBOLA case instance in a higher place, authors can extract the following information: name of authors, state of patients, year of publication, written report design (case written report, cohort report, or clinical trial or RCT), sample size, the infected point of time subsequently EBOLA infection, follow-up interval after vaccination time, efficacy, rubber, adverse furnishings after vaccinations, and QA canvass (Additional file six: Information S1).
Data checking
Due to the expected human error and bias, we recommend a data checking step, in which every included article is compared with its counterpart in an extraction sheet past evidence photos, to detect mistakes in information. We advise assigning articles to 2–3 independent reviewers, ideally not the ones who performed the extraction of those articles. When resources are limited, each reviewer is assigned a different article than the ane he extracted in the previous stage.
Statistical analysis
Investigators use different methods for combining and summarizing findings of included studies. Before assay, there is an of import step called cleaning of data in the extraction canvas, where the analyst organizes extraction sheet data in a class that tin can be read by analytical software. The analysis consists of 2 types namely qualitative and quantitative analysis. Qualitative analysis generally describes data in SR studies, while quantitative analysis consists of two main types: MA and network meta-analysis (NMA). Subgroup, sensitivity, cumulative analyses, and meta-regression are appropriate for testing whether the results are consistent or not and investigating the effect of certain confounders on the outcome and finding the best predictors. Publication bias should be assessed to investigate the presence of missing studies which can impact the summary.
To illustrate basic meta-analysis, we provide an imaginary information for the enquiry question about Ebola vaccine safety (in terms of adverse events, 14 days after injection) and immunogenicity (Ebola virus antibodies rise in geometric hateful titer, 6 months after injection). Bold that from searching and data extraction, we decided to do an analysis to evaluate Ebola vaccine "A" safety and immunogenicity. Other Ebola vaccines were not meta-analyzed because of the express number of studies (instead, it will exist included for narrative review). The imaginary information for vaccine safety meta-analysis tin can be accessed in Additional file 7: Data S2. To do the meta-analysis, we can apply free software, such as RevMan [22] or R packet meta [23]. In this example, we will use the R package meta. The tutorial of meta packet tin can be accessed through "General Parcel for Meta-Analysis" tutorial pdf [23]. The R codes and its guidance for meta-analysis done can exist found in Boosted file 5: File S3.
For the analysis, we assume that the study is heterogenous in nature; therefore, we cull a random effect model. Nosotros did an analysis on the safety of Ebola vaccine A. From the data tabular array, we tin run into some adverse events occurring after intramuscular injection of vaccine A to the subject of the written report. Suppose that we include six studies that fulfill our inclusion criteria. We tin can do a meta-analysis for each of the adverse events extracted from the studies, for example, arthralgia, from the results of random consequence meta-analysis using the R meta package.
From the results shown in Additional file three: Figure S3, we can see that the odds ratio (OR) of arthralgia is 1.06 (0.79; 1.42), p value = 0.71, which means that there is no clan betwixt the intramuscular injection of Ebola vaccine A and arthralgia, as the OR is almost one, and also, the P value is insignificant equally it is > 0.05.
In the meta-assay, nosotros can also visualize the results in a woods plot. Information technology is shown in Fig. three an example of a woods plot from the faux analysis.
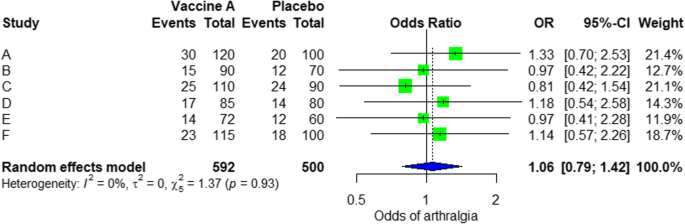
Random effect model forest plot for comparing of vaccine A versus placebo
From the forest plot, we tin can see 6 studies (A to F) and their corresponding OR (95% CI). The green box represents the effect size (in this example, OR) of each written report. The bigger the box means the study weighted more (i.eastward., bigger sample size). The blue diamond shape represents the pooled OR of the six studies. We can see the blue diamond cross the vertical line OR = 1, which indicates no significance for the association as the diamond nearly equalized in both sides. We tin confirm this besides from the 95% confidence interval that includes one and the p value > 0.05.
For heterogeneity, we run into that I 2 = 0%, which ways no heterogeneity is detected; the report is relatively homogenous (information technology is rare in the existent report). To evaluate publication bias related to the meta-analysis of adverse events of arthralgia, we can use the metabias role from the R meta parcel (Additional file iv: Figure S4) and visualization using a funnel plot. The results of publication bias are demonstrated in Fig. four. We run across that the p value associated with this test is 0.74, indicating symmetry of the funnel plot. We can confirm it past looking at the funnel plot.
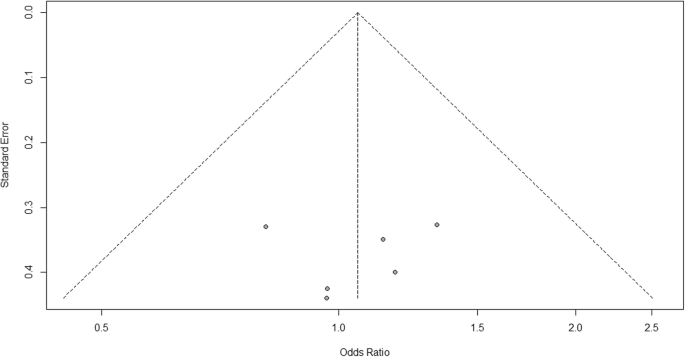
Publication bias funnel plot for comparison of vaccine A versus placebo
Looking at the funnel plot, the number of studies at the left and right side of the funnel plot is the same; therefore, the plot is symmetry, indicating no publication bias detected.
Sensitivity analysis is a procedure used to discover how different values of an contained variable volition influence the significance of a detail dependent variable past removing one report from MA. If all included study p values are < 0.05, hence, removing any written report will not change the significant association. It is but performed when there is a significant clan, then if the p value of MA done is 0.7—more than than one—the sensitivity analysis is not needed for this instance study example. If there are 2 studies with p value > 0.05, removing any of the two studies will upshot in a loss of the significance.
Double data checking
For more assurance on the quality of results, the analyzed data should be rechecked from full-text data by testify photos, to allow an obvious check for the PI of the study.
Manuscript writing, revision, and submission to a journal
Writing based on four scientific sections: introduction, methods, results, and discussion, generally with a conclusion. Performing a characteristic table for study and patient characteristics is a mandatory step which can exist constitute as a template in Additional file 5: Table S3.
After finishing the manuscript writing, characteristics table, and PRISMA flow diagram, the squad should send information technology to the PI to revise it well and reply to his comments and, finally, cull a suitable journal for the manuscript which fits with considerable bear upon factor and fitting field. We need to pay attention by reading the author guidelines of journals before submitting the manuscript.
Word
The role of evidence-based medicine in biomedical research is chop-chop growing. SR/MAs are also increasing in the medical literature. This paper has sought to provide a comprehensive arroyo to enable reviewers to produce loftier-quality SR/MAs. We hope that readers could gain general knowledge about how to conduct a SR/MA and have the conviction to perform ane, although this kind of written report requires complex steps compared to narrative reviews.
Having the bones steps for conduction of MA, at that place are many advanced steps that are applied for certain specific purposes. Ane of these steps is meta-regression which is performed to investigate the association of whatever confounder and the results of the MA. Furthermore, there are other types rather than the standard MA like NMA and MA. In NMA, we investigate the divergence between several comparisons when there were not enough data to enable standard meta-assay. It uses both straight and indirect comparisons to conclude what is the best between the competitors. On the other hand, mega MA or MA of patients tend to summarize the results of independent studies by using its individual subject area data. As a more detailed assay tin can be done, information technology is useful in conducting repeated measure assay and time-to-event analysis. Moreover, it tin perform analysis of variance and multiple regression analysis; however, it requires homogenous dataset and information technology is time-consuming in conduct [24].
Conclusions
Systematic review/meta-analysis steps include development of inquiry question and its validation, forming criteria, search strategy, searching databases, importing all results to a library and exporting to an excel canvass, protocol writing and registration, title and abstract screening, total-text screening, manual searching, extracting information and assessing its quality, data checking, conducting statistical analysis, double data checking, manuscript writing, revising, and submitting to a journal.
Availability of data and materials
Not applicative.
Abbreviations
- NMA:
-
Network meta-analysis
- PI:
-
Principal investigator
- PICO:
-
Population, Intervention, Comparison, Event
- PRISMA:
-
Preferred Reporting Items for Systematic Review and Meta-analysis statement
- QA:
-
Quality assessment
- SPIDER:
-
Sample, Phenomenon of Involvement, Pattern, Evaluation, Inquiry type
- SR/MAs:
-
Systematic review and meta-analyses
References
-
Bello A, Wiebe N, Garg A, Tonelli M. Evidence-based decision-making 2: systematic reviews and meta-assay. Methods Mol Biol (Clifton, NJ). 2015;1281:397–416.
-
Khan KS, Kunz R, Kleijnen J, Antes K. Five steps to conducting a systematic review. J R Soc Med. 2003;96(3):118–21.
-
Rys P, Wladysiuk M, Skrzekowska-Baran I, Malecki MT. Review manufactures, systematic reviews and meta-analyses: which can be trusted? Polskie Archiwum Medycyny Wewnetrznej. 2009;119(3):148–56.
-
Higgins JPT, Greenish S. Cochrane Handbook for Systematic Reviews of Interventions Version v.1.0 [updated March 2011]. 2011.
-
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339:b2535.
-
Methley AM, Campbell S, Chew-Graham C, McNally R, Cheraghi-Sohi South. PICO, PICOS and SPIDER: a comparing report of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv Res. 2014;14:579.
-
Gross L, Lhomme Eastward, Pasin C, Richert Fifty, Thiebaut R. Ebola vaccine development: systematic review of pre-clinical and clinical studies, and meta-analysis of determinants of antibody response variability afterward vaccination. Int J Infect Dis. 2018;74:83–96.
-
Shea BJ, Reeves BC, Wells Chiliad, Thuku M, Hamel C, Moran J, ... Henry DA. AMSTAR 2: a disquisitional appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ. 2017;358:j4008.
-
Giang HTN, Banno Thou, Minh LHN, Trinh LT, Loc LT, Eltobgy A, et al. Dengue hemophagocytic syndrome: a systematic review and meta-assay on epidemiology, clinical signs, outcomes, and risk factors. Rev Med Virol. 2018;28(6):e2005.
-
Morra ME, Altibi AMA, Iqtadar S, Minh LHN, Elawady SS, Hallab A, et al. Definitions for warning signs and signs of severe dengue according to the WHO 2009 classification: systematic review of literature. Rev Med Virol. 2018;28(4):e1979.
-
Morra ME, Van Thanh L, Kamel MG, Ghazy AA, Altibi AMA, Dat LM, et al. Clinical outcomes of current medical approaches for Eye East respiratory syndrome: a systematic review and meta-analysis. Rev Med Virol. 2018;28(3):e1977.
-
Vassar M, Atakpo P, Kash MJ. Transmission search approaches used by systematic reviewers in dermatology. Journal of the Medical Library Association: JMLA. 2016;104(four):302.
-
Naunheim MR, Remenschneider AK, Scangas GA, Bunting GW, Deschler DG. The effect of initial tracheoesophageal vocalisation prosthesis size on postoperative complications and vocalization outcomes. Ann Otol Rhinol Laryngol. 2016;125(half dozen):478–84.
-
Rohatgi AJaiWa. Web Plot Digitizer. ht tp. 2014;2.
-
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5(1):13.
-
Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard difference from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;xiv(1):135.
-
Van Rijkom HM, Truin GJ, Van't Hof MA. A meta-analysis of clinical studies on the caries-inhibiting upshot of fluoride gel handling. Carries Res. 1998;32(2):83–92.
-
Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman Ad, et al. The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928.
-
Tawfik GM, Tieu TM, Ghozy S, Makram OM, Samuel P, Abdelaal A, et al. Speech communication efficacy, safety and factors affecting lifetime of voice prostheses in patients with laryngeal cancer: a systematic review and network meta-analysis of randomized controlled trials. J Clin Oncol. 2018;36(15_suppl):e18031-east.
-
Wannemuehler TJ, Lobo BC, Johnson JD, Deig CR, Ting JY, Gregory RL. Vibratory stimulus reduces in vitro biofilm formation on tracheoesophageal voice prostheses. Laryngoscope. 2016;126(12):2752–vii.
-
Sterne JAC, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing gamble of bias in non-randomised studies of interventions. BMJ. 2016;355.
-
RevMan The Cochrane Collaboration %J Copenhagen TNCCTCC. Review Manager (RevMan). v.0. 2008.
-
Schwarzer GJRn. meta: An R package for meta-analysis. 2007;7(three):40-45.
-
Simms LLH. Meta-analysis versus mega-analysis: is there a departure? Oral budesonide for the maintenance of remission in Crohn's affliction: Faculty of Graduate Studies, University of Western Ontario; 1998.
Funding
This study was conducted (in part) at the Joint Usage/Enquiry Eye on Tropical Disease, Institute of Tropical Medicine, Nagasaki University, Nihon.
Author data
Affiliations
Contributions
NTH and GMT were responsible for the idea and its design. The figure was done by GMT. All authors contributed to the manuscript writing and approval of the concluding version.
Corresponding author
Ideals declarations
Ethics approval and consent to participate
Not applicative.
Consent for publication
Non applicative.
Competing interests
The authors declare that they have no competing interests.
Additional data
Publisher's Notation
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Boosted files
Boosted file 1:
Figure S1. Hazard of bias assessment graph of included randomized controlled trials. (TIF 20 kb)
Boosted file 2:
Figure S2. Risk of bias assessment summary. (TIF 69 kb)
Additional file three:
Figure S3. Arthralgia results of random effect meta-analysis using R meta parcel. (TIF twenty kb)
Additional file 4:
Figure S4. Arthralgia linear regression test of funnel plot asymmetry using R meta package. (TIF xiii kb)
Additional file 5:
Table S1. PRISMA 2009 Checklist. Table S2. Manipulation guides for online database searches. Table S3. Detailed search strategy for twelve database searches. Tabular array S4. Baseline characteristics of the patients in the included studies. File S1. PROSPERO protocol template file. File S2. Extraction equations that can be used prior to analysis to become missed variables. File S3. R codes and its guidance for meta-analysis done for comparison between EBOLA vaccine A and placebo. (DOCX 49 kb)
Boosted file half dozen:
Data S1. Extraction and quality cess data sheets for EBOLA case example. (XLSX 1368 kb)
Additional file vii:
Information S2. Imaginary data for EBOLA example example. (XLSX ten kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Eatables Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in whatsoever medium, provided yous requite appropriate credit to the original author(southward) and the source, provide a link to the Artistic Eatables license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/cypher/1.0/) applies to the data made available in this article, unless otherwise stated.
Reprints and Permissions
Almost this commodity
Cite this article
Tawfik, G.G., Dila, K.A.S., Mohamed, One thousand.Y.F. et al. A step past step guide for conducting a systematic review and meta-analysis with simulation information. Trop Med Wellness 47, 46 (2019). https://doi.org/10.1186/s41182-019-0165-six
-
Received:
-
Accustomed:
-
Published:
-
DOI : https://doi.org/10.1186/s41182-019-0165-vi
Keywords
- Search
- Data
- Extraction
- Analysis
- Study
- Results
hernandezbetalor1988.blogspot.com
Source: https://tropmedhealth.biomedcentral.com/articles/10.1186/s41182-019-0165-6
0 Response to "I Conducted a Search for Meta-analyses and Systematic Reviews"
Post a Comment